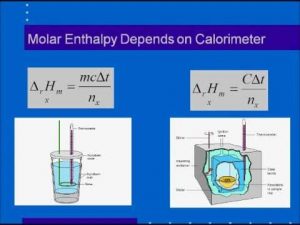
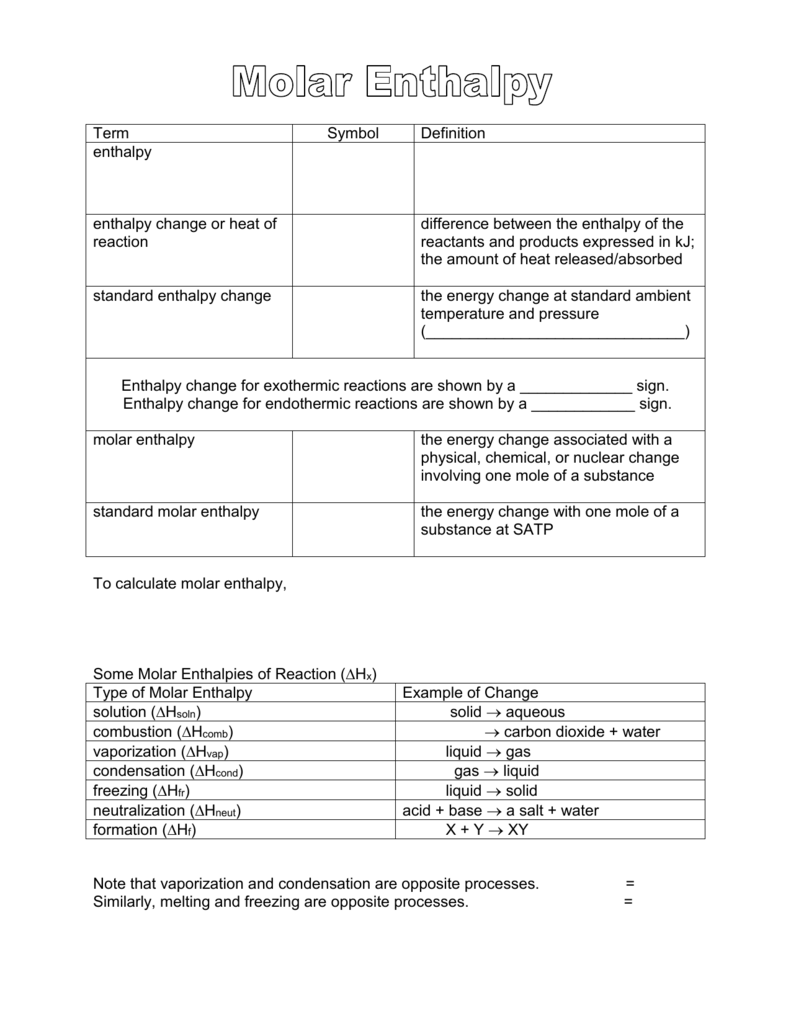
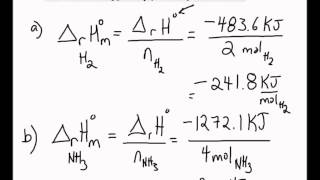SOLVED:3. Given the molar enthalpy changes for the reactions below calculate the enthalpy of formation of zinc oxide_ Zno) + 2Ht(aq) ~> Zn?t(aq) + Hz (g) AH=-151 kJlmol ZnOts) + 2Ht(aq) ~>

Calculate the change in molar entropy and change in Gibbs' free energy when 1 mol of liquid butanol vaporizes at 25ºC to a gas that is at 0.001 atm? | Socratic

3:04 calculate the molar enthalpy change (ΔH) from the heat energy change, Q - TutorMyself Chemistry

SOLVED:The molar enthalpy change for the neutralization reaction: HCI (aq) + NaOH (aq) +> NaCl (aq) + HzO (I) is AH=-57.3 k/mol. This is an exothermic reaction which means heat is released.